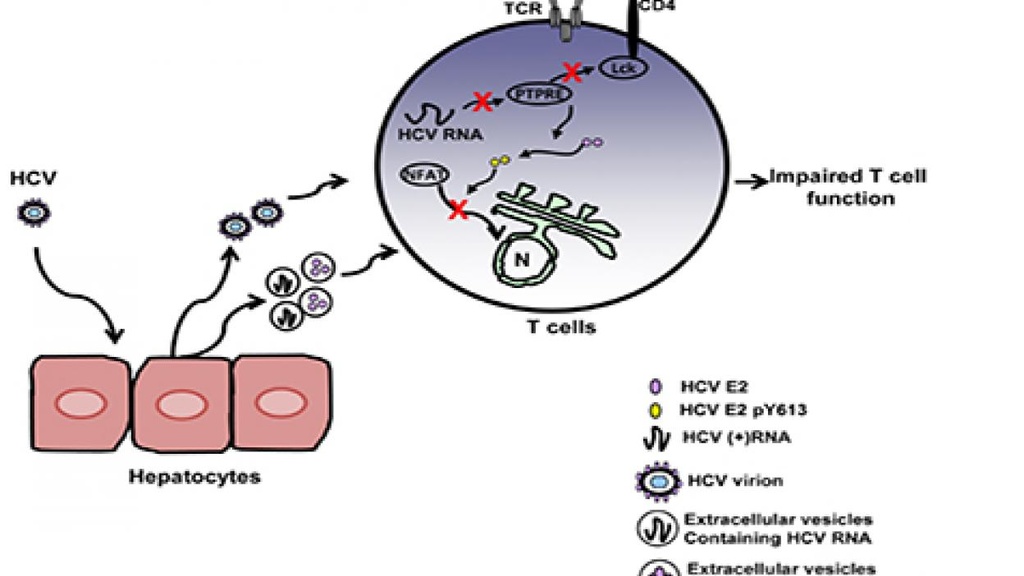
Proposed Model
Proposed model for inhibition of T cell receptor (TCR) signaling during HCV infection.
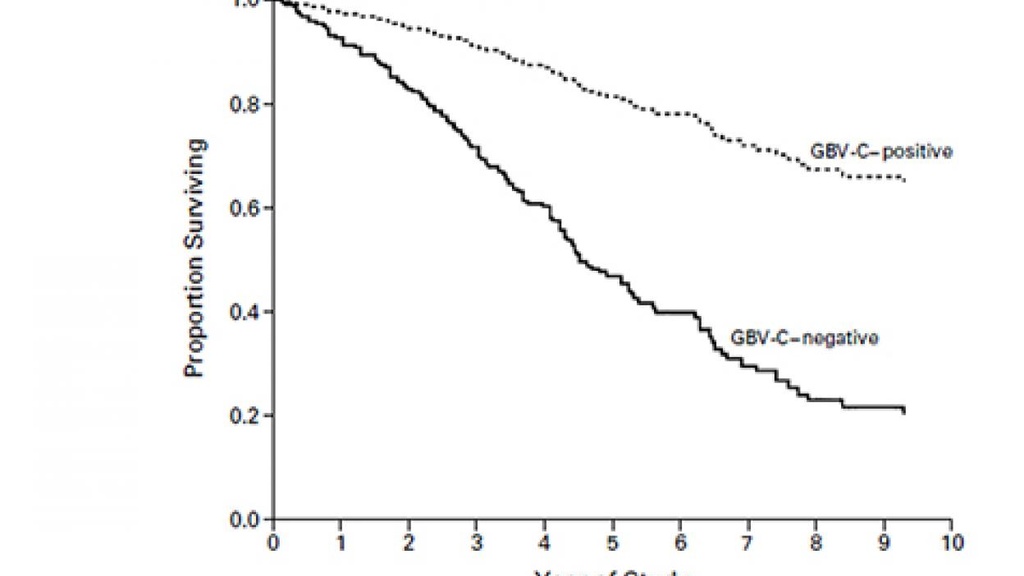
Key Finding
Survival Curves for HIV-Infected Patients with and Patients without GBV-C Viremia.
Welcome to the Stapleton Lab
Our laboratory investigates the interactions between human flaviviruses (HCV, GBV-C, YFV, Dengue, Zika) and human T cells. We also examine the relationships between GBV-C and HIV. The group studies the effects of these viruses on clinical outcomes and host immune responses in both translational experiments (using fresh and stored clinical specimens), and works to elucidate the mechanisms that contribute to the findings we observe.
Earlier work in the lab demonstrated that GBV-C inhibits HIV replication and that two GBV-C proteins interfere with HIV entry, and that GBV-C infection reduces ex vivo T cell activation, proliferation and HIV reactivation. A motif within the envelope protein E2 (aa 276-292) directly blocks HIV entry while a different E2 region (aa 82-98) inhibits signaling through the T cell and IL-2 receptors by competing with the tyrosine kinase (Lck), blocking signaling by both receptors. Recent studies found that HCV and YFV also inhibit TCR signaling by two independent mechanisms. First, the viral envelope protein is phosphorylated by Lck, and phospho-E2 inhibits NFAT translocation into the nucleus, blunting distal signaling through the TCR and reducing IL-2 production. Secondly, a short, viral-genome derived RNA inhibits proximal TCR signaling by reducing the expression of a Src kinase regulatory phosphatase PTPRE. These immune modulating motifs have two potential translational opportunities. The first is to modify the viral envelope to abolish the T cell inhibitory effect, thus making the envelope proteins more immunogenic. The second is to interfere with the immunogenicity, testing this in gene delivery systems to determine if we can reduce immunogenicity of heterologous proteins. This may also be useful in immune-mediated disease states and the present focus of the lab.




